Early Spring Events
Our Early Spring Events Here’s a slightly longer version with the events included: This early spring, you’ll find us at several exciting events ac...
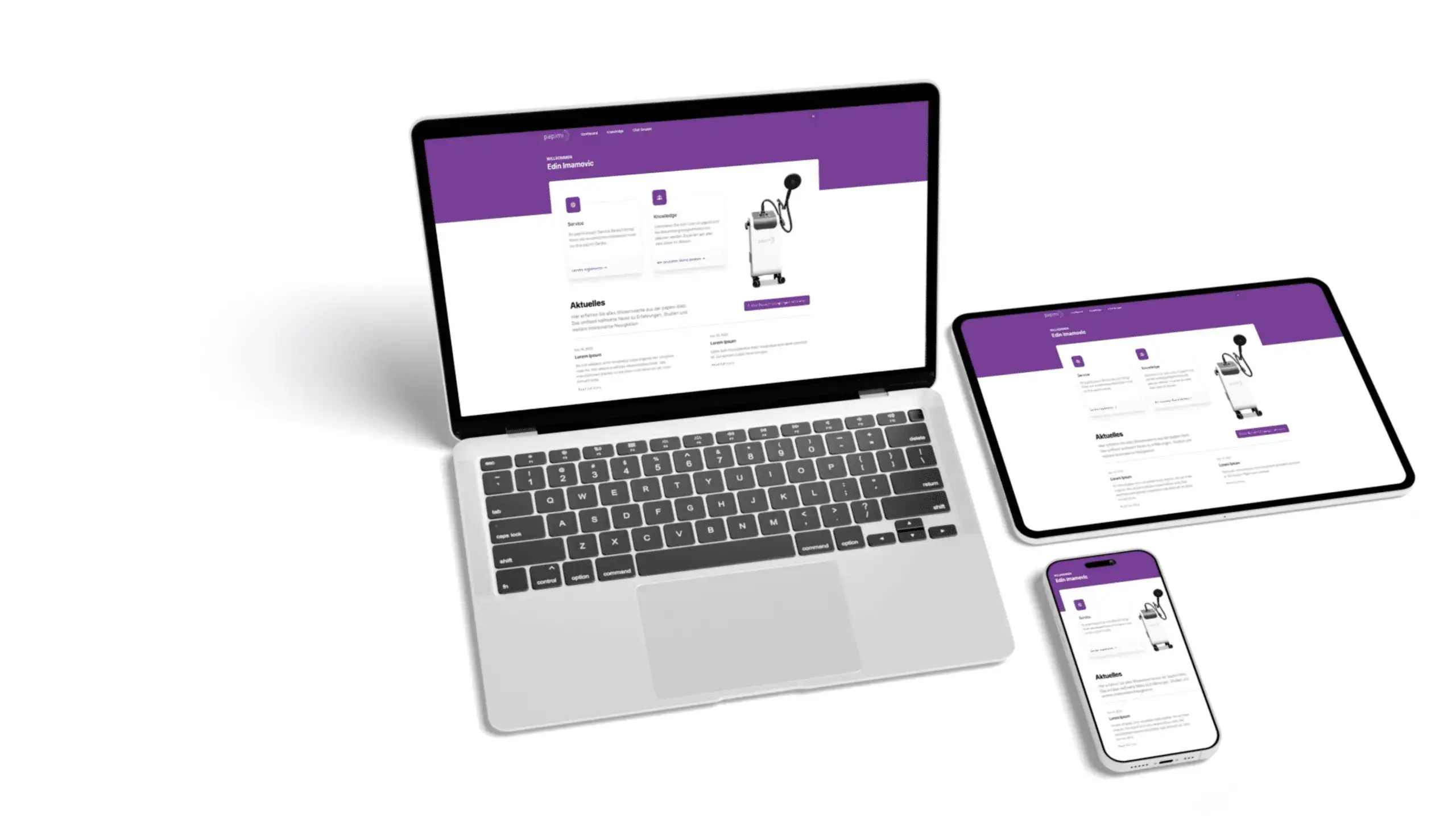
The device as well as the production are certified according to the EC guidelines 93/42/EEC Annex II as well as EN ISO 13485.


papimi is a certified and approved medical device of class IIa.
The American patent was granted on September 17, 1996 (US 5,556,418). The first European patent (EP 0648.140.B1) was granted on January 31, 2001 and the second (EP 04.743.704.1) in August 2003.
Our premium service ensures that your device always performs flawlessly. Our staff will also inform you about our many warranty options.
The device as well as the production are certified according to the EC guidelines 93/42/EEC Annex II as well as EN ISO 13485.


papimi is a certified and approved medical device of class IIa.
The American patent was granted on September 17, 1996 (US 5,556,418). The first European patent (EP 0648.140.B1) was granted on January 31, 2001 and the second (EP 04.743.704.1) in August 2003.
Our premium service ensures that your device always performs flawlessly. Our staff will also inform you about our many warranty options.
With the papimi DELTA, Ion-Induction-Therapy (IIT) has arrived in the 21st century. This Physical Cell Therapy sets new standards in medicine. Learn more about the Original by Prof. DDr. Pappas.

Spiral-applicator XL
Ring-applicator
Spiral-applicator M
Ion-Induction-Device with applicator-arm

With the papimi DELTA, Ion-Induction-Therapy (IIT) has arrived in the 21st century. This Physical Cell Therapy sets new standards in medicine. Learn more about the original by Prof. DDr. Pappas.













Clinical Trial Confirms: papimi Reduces Back Pain by More Than

EffectiveBioMedicine Congress in Vienna 2025 We are incredibly proud to

Case Report: Refractory Seizures Controlled with papimi IIT in Patient
Our Early Spring Events Here’s a slightly longer version with the events included: This early spring, you’ll find us at several exciting events ac...
Clinical Trial Confirms: papimi Reduces Back Pain by More Than 50% in Just 3 Sessions Clinical Trial Confirms: papimi Reduces Back Pain by More Than 5...
EffectiveBioMedicine Congress in Vienna 2025 We are incredibly proud to have been part of the Effective BioMedicine Congress 2025 – not only as ...
Case Report: Refractory Seizures Controlled with papimi IIT in Patient with Benign Brain Tumor A 39-year-old woman with a non-operable lipoma in the c...
papimi IIT & Prostate Health: What Clinical and Practical Experience Show Can a non-invasive, painless method offer support for men with benign pr...
New Study: papimi Reduces Pain and Balances Nervous System — After Just One Session 🧬 New Study: papimi Reduces Pain and Balances Nervous System ...
The papimi technicians team welcomes three new experts from Croatia and the UK! Fully trained, they are now providing top-level support worldwide....
New specialist article available: How is Ion-Induction-Therapy (IIT) with papimi transforming modern dentistry? Find out in "OM & Nutrition 2024." Rea...
NieCo 2024 exceeded expectations with its stellar lineup of speakers, engaging seminars, hands-on workshops, and a fantastic venue, making it an aweso...
2023 completed We will be back on the 8th of January 2024 (For technical support during this time, please call our headquarters in Athens directly: +3...

Learn how papimi is used in modern practice settings. During a no-obligation consultation, we’ll answer your questions and share insights from over 30 years of professional experience.

Learn how papimi is used in modern practice settings. During a no-obligation consultation, we’ll answer your questions and share insights from over 30 years of professional experience.
